Participate - University Hospitals Birmingham NHS Foundation Trust
This site is not currently recruiting
This site is recruiting participants from the West Midlands area.
Comparing COVID-19 Booster Vaccinations (COV-BOOST) - Young Adults Fractional Dosing Sub-Study
What is the purpose of this research trial?
There are now a number of vaccines that have been approved in the UK to prevent COVID-19 and other vaccines that are still in UK clinical trials, which may be approved later in the year. Millions of people have now received their first 2 vaccinations, which we call a “prime-boost” course. There were 3 vaccines used by the NHS to deliver an initial prime-boost during the first part of the NHS immunisation campaign: ChAdOx1-nCov19 (commonly known as the “Oxford vaccine” or ‘AstraZeneca Vaccine’), BNT162b2 (commonly known as the “Pfizer vaccine”) and mRNA-1273 (commonly known as the “Moderna vaccine”). The NHS has now recommended all adults receive a 3rd dose booster vaccine against COVID-19 at least 3 months after they received their second dose.
Young adults have a stronger immune response to vaccines than older adults, and results from the first stage of COV-BOOST and other COVID-19 vaccine studies have suggested that lower doses of BNT162b2 (Pfizer) or mRNA-1273 (Moderna) may give as good an immune response in young adults as higher doses. They may also be associated with fewer side effects or lower rates of already rare adverse events. Giving lower doses may also allow existing stocks of vaccines to be given to more people, which is important while need for the vaccine is greater than the number of doses available. This study is to evaluate the safety and side effect profile of giving lower doses of the Pfizer or Moderna COVID-19 vaccine to young adults, as well as assessing its impact on the immune response to the virus which causes COVID-19, called SARS-CoV-2.
We will be enrolling adults aged 18 to 30 years old who have had 2 doses of BNT162b2 (Pfizer) or mRNA-1273 (Moderna), are at least 3 months (84 days) after their second dose and have not yet had a booster.
What are the vaccines against?
These vaccines are against the coronavirus SARS-CoV2 that causes the disease COVID-19.
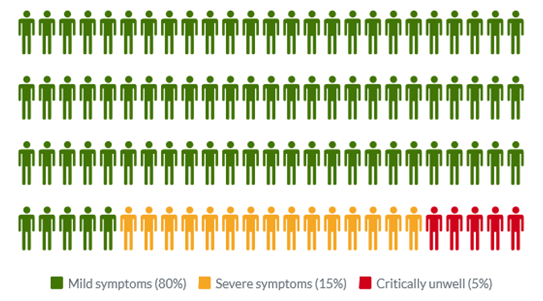
Common symptoms of COVID-19 include fever, tiredness, dry cough, and changes to taste and smell. Whilst about 80% of infected people have no or mild symptoms and will recover from the infection without needing special treatment, approximately 10-15% of cases (2-3 in 20) progress to develop severe symptoms, and about 5% (1 in 20) become critically ill.
There are some treatments that have been shown to be effective in reducing the severity of disease and the risk of death; but at present there is no cure. Older people and those with underlying medical conditions are more likely to develop serious illness. It has also been seen that people of some ethnic groups (Black and Asian) might be at a greater risk of severe illness. Some people also have symptoms that last a long time after they have recovered (commonly referred to as “long-COVID”). This is why effective vaccines are so important.
Am I suitable to take part?
Adults who are aged 18 – 30 years who have had 2 doses of BNT162b2 (Pfizer) or mRNA-1273 (Moderna) and are at least 3 months (84 days) since their second dose are eligible to take part, providing that they have not yet received a booster. In order to participate in the study:
- You must be willing to tell the trial staff about your medical history, and you may be asked to allow the trial staff to check this with your General Practitioner (GP). Bear in mind that we would also notify your GP if you joined the trial (even if we did not need to check your medical history with them in advance).
- You must agree not to donate blood during the trial
For further details on why you could not take part in this study please see the Participant Information Sheet .
Summary of the study
Participants will be randomised to receive either a single dose of the BNT162b2 (Pfizer) vaccine (30mcg), one third of the normal dose of BNT162b2 (10mcg, the same dose being used in other countries for children aged 5 – 11 years), a half dose of the mRNA-1273 (Moderna) vaccine (50mcg, the dose being used by the NHS as a 3rd dose booster) or one quarter of a dose of the mRNA-1273 vaccine (25mcg). They will then complete a diary of their symptoms over the next 7 days, and any other adverse events which occur up to 3 months after having had the vaccine. There are several follow up visits to have blood tests to check for immune markers, and to check on the participants health.
Participants will initially be “blinded”, meaning they will not be told which vaccine they have been given until after they have attended a follow up visit 84 days after being vaccinated. If a participant is travelling prior to their day 84 follow up visit it may be possible to unblind them before the day 84 follow up visit if there is an urgent need and they have attended the follow up visit that is 28 days after vaccination.
What vaccines are being used in this study?
| BNT162b2 (Pfizer/BioNTech) 30mcg |
| BNT162b2 (Pfizer/BioNTech) 10mcg |
| mRNA-1273 (Moderna) 50mcg |
| mRNA-1273 (Moderna) 25mcg |
BNT162b2 (Pfizer/BioNTech): Common use under emergency provision
This is the vaccine commonly known as ‘The Pfizer vaccine.’ This is a messenger RNA (mRNA) vaccine. This vaccine uses a small amount of the genetic coding material (mRNA) of the SARS-CoV-2 spike (S) protein packaged inside very small fatty particles (lipid nanoparticles). When these are injected into your body, your cells take up these fatty particles, and then start producing the SARS-CoV-2 spike protein. Your immune system then “sees” these spike proteins and makes a protective immune reaction against them. The original mRNA that has been taken into your cells is broken down within a few days and cannot be incorporated into your own genetic code.
This vaccine has been tested in more than 40,000 people worldwide in clinical trials and subsequently given to tens of millions of people, and has been shown to be both safe, and effective.
The vaccine does not contain a live version of the SARS-CoV-2 coronavirus and therefore cannot give you COVID-19. The potential side effects of these vaccines are discussed in more detail in the section ‘What are the risks of taking part in this trial’.
mRNA-1273 (Moderna): Common use under emergency provision
This is the vaccine commonly known as “The Moderna vaccine”. This is also a messenger RNA (mRNA) vaccine which works in a similar way to BNT162b2 (The Pfizer vaccine).
This vaccine has been tested in more than 30,000 people worldwide in clinical trials and subsequently given to tens of millions of people, has been shown to be both safe, and effective.
Participants will not know which vaccine or dose they have received until 84 days after they have received the vaccine.
What are the side effects of these vaccines?
Common side effects
People very often have tenderness, pain, warmth, redness, itching, swelling or bruising or less commonly have a small lump in their arm where they have been vaccinated.
Other common systemic side effects
Some people can develop these symptoms after vaccination. They usually last for less than a week after you are vaccinated (more commonly 24-48hours afterwards).
- Fatigue
- Headaches
- Flu-like symptoms, such as high temperature, sore throat, runny nose, cough and chills
- Muscle aches
- Joint aches
- Feeling unwell (malaise)
- Feeling sick or nauseated or vomiting
Other less common side effects:
- Abdominal pain
- Decreased appetite
- Feeling dizzy
- Swollen lymph nodes (glands)
- Excessive sweating, itching skin or rash
These symptoms can be reduced by use of paracetamol around the time of immunisation and over the next 24 hours.
After immunisation with the BNT162b2 (Pfizer/BioNTech) vaccine, difficulty sleeping has been observed in fewer than 1 in 100 people, and weakness of the muscles on one side of the face has been observed in fewer than 1 in 1000 people.
What are the advantages of taking part?
We anticipate that participating in the trial will mean that you gain some additional protection against the coronavirus. Most importantly, the information gained from the trial will make a valuable contribution to the pandemic response.
Are there any risks from taking part in the study?
In addition to the potential side effects of the vaccines outlined above, the blood samples taken in the study may cause slight pain and occasionally bruising. Please refer to the participant information sheet for full details of procedures and potential risks.
What will happen if I don’t want to carry on with the trial?
If, at any time, after enrolment, you change your mind about being involved with this trial you are free to withdraw without giving a reason. If you withdraw we would not usually perform any more research procedures; although occasionally we might need to offer you a follow up visit for safety purposes, for example for blood tests. You would not have to agree to this. Your decision will not result in any penalty. Unless you state otherwise, any samples taken whilst you have been in the trial will continue to be stored and used for research as detailed above. You are free to request that your samples are destroyed at any time during or after the trial. Your data would be managed as laid out in the section ‘What will happen to my data’. If you choose to withdraw from the trial, your standard medical care will not be affected.
What’s next?
If you would like to find out more, please read the Participant Information Sheet and if you are interested in taking part, please complete our pre-screening questionnaire.
Download the Participant Information Sheet (PDF)
This site is not currently recruiting
Complete the Pre-screening Questionnaire
For further details contact us on :
contact details will follow shortly




